PRODUCT IDENTIFICATION
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
REFRACTIVE INDEX
150 C
APPLICATIONS
Benzoic acid derivatives substituted by hydroxy group or ether containing oxygen atom have active bacteriostatic and fragrant properties. They are typically used in pharmaceutical and perfumery industry. The destructive metabolic property of oxygen containing benzoic acid derivatives such as protocatechuic acid (3,4-dihydroxybenzoic acid) and veratric acid (3,4-dimethoxybenzoic acid) is used in the application for pharmaceuticals. Protocatechuic acid is a catabolite of epinephrine. Prazosin, a peripheral vasodilator and antihypertensive, is also an example of the applicaion of veratric acid. Hydroxy and ether substituted benzoic acids feature analogue metabolite of aspirin (acetylsalicylic Acid). They are used as intermediates for pharmaceuticals (especially for antipyetic anlgesic, antirheumatism) and other organic synthesis. They are used as matrix for ionization of peptides, proteins and carbohydrates.
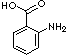
Benzoic acid, the simplest aromatic carboxylic acid containing carboxyl group bonded directly to benzene ring, is a white, crystalline organic compound; melting at 122 C (starting sublime at 100 C); boiling at 249 C; slightly soluble in water, soluble in ethanol, very slightly soluble in benzene and acetone. Its aqua solution is weakly acidic. It occurs naturally in many plants and resins. Benzoic acid is also detected in animals. The most of commercial benzoic acid is produced by the reaction of toluene with oxygen at temperatures around 200 C in the liquid phase and in the presence of cobalt and manganese salts as catalysts. It can be prepared also by the oxidation of benzene with concentrated sulphuric acid or carbon dioxide in the presence of catalysts. Other methods are such as by the oxidation of benzyl alcohol, benzaldehyde, cinnamic acid; by hydrolysis of benzonitrile, benzoyl chloride. More than 90% of commercial benzoic acid is converted directly to phenol and caprolactam. Its use in the production of glycol benzoates for the application of plasticizer in adhesive formulations is increasing. It is also used in the manufacture of alkyd resins and drilling mud additive for crude oil recovery applications. It is used as a rubber polymerization activators and retardants. Benzoic acid is converted to its salts and esters for the use of preservative application in foods, drugs and personal products. Sodium benzoate, sodium salt of benzoic acid, is used preferably as one of the principal anti-microbial preservatives used in foods and beverages (but it's concentration is limited usually not exceeding 0.1% because it is poisonous), as it is about 200 times more soluble than benzoic acid. Sodium Benzoate is also used in medications, anti-fermentation additives and tabletting lubricant for pharmaceuticals. The industrial applications are as a corrosion inhibitor, as an additive to automotive engine antifreeze coolants and in other waterborne systems, as a nucleating agents for polyolefin, as a dye intermediate, as a stabilizer in photographic processing and as a catalyst. Wide range of benzoic esters are used as solvents, dying carrier, disinfectant additive, penetrating agent and pesticides and manufacturing other compounds.Anthranilic acid, amino group substituted benzoic acid at ortho position, is used as an intermediate for production of dyes, pigments and saccharin. It has amino and carboxylic group attached in ring structure. It and its esters are used in preparing perfumes, pharmaceuticals and UV-absorber as well as corrosion inhibitors for metals and mold inhibitors in soya sauce.
APPEARANCE
99.0% min
ASH
0.2% max
MOISTURE
0.5% max
MELTING POINT
146 ± 1.5 C
OTHER
INFORMATION