PRODUCT IDENTIFICATION
110-89-4
EINECS NO.
203-813-0
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
REFRACTIVE INDEX
15 C
GENERAL DESCRIPTION & APPLICATIONS
Piperidine, hexahydropyridine, is a family of heterocyclic organic compound derived from pyridine through hydrogenation. It has one nitrogen atom in the cycle. It is a clear liquid with pepper-like aroma. It boils at 106 C, soluble in water, alcohol, and ether. The major application of piperidine is for the production of dipiperidinyl dithium tetrasulfide used as a rubber vulcanization accelerator. In pharmaceutical synthesis industry, it is a skeleton in some drugs such as methylphenidate (central nervous system stimulant), budipine (antiparkinsonian drug) raloxifene (selective estrogen receptor modulator), minoxidil (an oral drug to treat high blood pressure). It is used as a special solvent in solid phase synthesis and a protecting group for peptide synthesis. Piperidine derivative compounds are used as intermediate to make crystal derivative of aromatic nitrogen compounds containing nuclear halogen atoms. Ring system compounds with nitrogen which have basic property playing important roles as cyclic component in industrial field such as raw materials for hardener of epoxy resins, corrosion inhibitors, insecticides, accelerators for rubber, urethane catalysts, antioxidants and as a catalyst for silicone esters. They are used in manufacturing pharmaceuticals. Piperidine is listed as a Table II precursor under the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances.
APPEARANCE
99.5% min
COLOR, APHA
10 max
SPECIFIC GRAVITY
0.857 - 0.867
2401
Picoline :
Three structural isomers of methyl pyridines (alpha, beta, gamma-
positions)
Lutidine : Six structural isomers of dimethyl pyridines (2,3-,
,24-, 2,5-, 2,6-, 3,4-, 3,5- positions)
Collidine : Three structural isomers
of trimethyl pyridines (2,3,5-, 2,3,6-, 2,4,6- positions)
Pyrimidine:
Pyridine alteration containing nitrogen atoms at positions 1 and
3
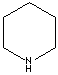
Piperidine: Hexahydropyridine (saturated form)
Nicotinic acid:
pyridine-3-carboxylic acid
Pyridine and its derivatives are very important in industrial field as well as in bio chemistry. Nucleotide consist of either a nitrogenous heterocyclic base (purine or pyrimidine). Three major pyrimidines in living systems are cytosine, thymine, and uracil. Pyrimidine and its derivatives are biologically important components of nucleic acids (DNA, RNA) and coenzymes. Some pyridine system is active in the metabolism in the body. Certain nitrogenous plant products also have pyridine class compounds. They can be the parent compound of many drugs, including the barbiturates. Pyridine and its derivatives are used as solvents and starting material for the synthesis of target compounds such as insecticides, herbicides, medicines, vitamins, food flavorings, feed additives, dyes, rubber chemicals, explosives, disinfectants, and adhesives. Pyridine is also used as a denaturant for antifreeze mixtures, as a dyeing assistant in textiles and in fungicides.
PRICE