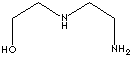
| AMINOETHYLETHANOLAMINE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. |
111-41-1 |
|
| EINECS NO. | 203-867-5 | |
| FORMULA | H2N(CH2)2NH(CH2)2OH | |
| MOL WT. | 104.15 | |
| H.S. CODE | 2922.19 | |
|
TOXICITY |
Oral, rat: LD50 = 3 gm/kg | |
| SYNONYMS | 2-(2-Aminoethylamino)-Ethanol; N-hydroxyethyl-1,2-ethanediamine; | |
| N-hydroxyethylethylenediamine; N-(2-Hydroxyethyl)ethylenediamine; 2-((aminoethyl)amino)ethanol; N-aminoethylethanolamine; | ||
|
SMILES |
|
|
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | colorless liquid | |
| MELTING POINT |
-18 C | |
| BOILING POINT | 140 C | |
| SPECIFIC GRAVITY | 1.03 | |
| SOLUBILITY IN WATER | slightly soluble | |
| AUTOIGNITION |
345 C | |
| VISCOSITY | ||
| pH |
| |
| VAPOR DENSITY | 3.59 | |
| NFPA RATINGS | Health: 2; Flammability: 1; Reactivity: 0 | |
|
REFRACTIVE INDEX |
1.4861 | |
| FLASH POINT |
129 C | |
| STABILITY | Stable under ordinary conditions | |
|
APPLICATIONS |
||
| Alkanolamines have the combined physical and chemical characteristics of both alcohols and amines in one molecule, which makes them useful intermediates in the synthesis of various target molecules for the use in many diverse areas such as pharmaceutical, urethane catalysts, coatings, personal care, products, water treatments, corrosion inhibitors, and gas treating industries. There are 1°, 2º or 3º nitrogen atom and one hydroxyl group at least in alkanolamines. Alkanolamines react with inorganic acids carboxylic acids to form salts, soaps, esters, or amides. Alkanolamines are used in both water- based and solvent-based coatings to enhance the solubility, reducibility, pigment dispersing and pH stability. They are used in cathodic electrodeposition systems and as a catalyst for chain-extend. Alkanolamines are used to prepare surface-active soaps through reaction with fatty acids. Surface-active soaps are used commercially as a emulsifier, lubricants, detergents, pesticides and personal care products. Alkanolamines maintain a constant alkalinity in the boiling water flows and condensate not to form solid products which would impede line flow. This function is applied for corrosion Inhibits. Alkanolamines are widely employed in the preparation of water soluble cationic flocculants and ion exchange resins which adsorb solid and colloidal particles by electrostatic attraction. They are used for water treatment industry. Alkanolamines and their derivatives are widely used as intermediates for the production of active pharmaceutical ingredients such as procaine, antihistamines analgesics from N,N-dimethylethanolamine or N-methyldiethanolamine. Alkanolamines are used to remove hydrogen sulfide (H2S) and CO2 gas from gas streams in natural and refinery gas operations. Aminoethyl ethanolamine which has amine groups and hydroxyl group is used as an important intermediate for polymer condensation, pharmaceuticals, agrochemicals, paper chemicals, rubber chemicals, textile auxiliaries. Aminoethyl ethanolamine is used to produce shampoo, cationic surfactants, antistatic agents and chelating agents. | ||
| SALES SPECIFICATION | ||
|
APPEARANCE |
colorlessto pale yellow liquid | |
| ASSAY (G.C.) |
99.0% | |
| COLOR (APHA) |
20 max | |
| TRANSPORTATION | ||
| PACKING | 200kgs in Drum | |
| HAZARD CLASS | 8 (9.2) | |
| UN NO. | 2735, 1759 | |
GENERAL DESCRIPTION OF ETHYLENEDIAMINES |
||
|
Ethylenediamine is a colourless to yellowish, strongly alkaline liquid, melting
at 8.5 C, boiling at 116 C; completely soluble in water and soluble alcohol. It
is a manufactured chemical that does not occur naturally. It has two primary
amine groups. There are homologous odd number amine (on even number linear
carbon chain) series of ethylenediamines; diethylenetriamine (linear C-4
diamine), triethylenetetramine (linear C-6 triamine), tetraethylenepentamine
(linear C-8 pentamine), and pentaethylenehexamine (linear C-10 hexamine).
Diethylenediamine is the simplest cyclic ethyleneamine (C-4), called piperazine. It has two secondary amine groups in cyclic system. It is a deliquescent crystalline compound melting at 105 C; soluble in water, alcohol, glycerol, and glycols. It is used as a main ingredient of anthelmintics and psychoactive drugs. Aminoethylpiperazine is also a member of C- 6 cyclic ethyleneamine which has aminoethyl attached to a nitrogen in piperazine. Accordingly, it has one primary, one secondary, and one tertiary nitrogen atom. It is used in the synthesis of catalysts, epoxy curing agent, and corrosion inhibitors. Aminoethylethanolamine (AEEA) is an analogue of diethylenetriamine. A hydroxyl group replace for an primary amine group. AEEA has one primary amine, one secondary amine, and one primary hydroxyl group. AEEA is a useful intermediate in the production of surfactants, chelating agents, and curing agents. Other branched or cyclic ethylenediamines include N,N'-Bis-(2-aminoethyl)piperazine) [CAS #: 6531-38-0], N-[(2-aminoethyl)2-aminoethyl]piperazine) [CAS #: 24028-46-4], tris(2-aminoethyl)amine) [CAS #: 4097-89-6]. Ethylenediamines are produced by the reaction of aqueous ammonia with 1,2-dichloroethane. This process yields the mixture of polyamines in the form of hydrochloride salts. The next step is neutralisation of the salts with aqueous caustic soda to separate free amines. The individual free amines are isolated by fractional distillation. Ethylenediamines are capable of entering into a variety of reactions because of their combination of reactivity, basicity, and surface activity and of the basic functionality of the nitrogen atom. They are therefore important intermediates for a wide variety of chemical syntheses. Examples of products obtained by reacting amines are as follows:
Ethylenediamine is used as a very important bidentate ligand forming chelate agents. The main application is to produce chelating agents such as ethylenediaminetetraacetic acid (EDTA). It is used in the manufacture of carbamate fungicides, surfactant and dyes. It is useful also in manufacturing accelerator or curing agent in epoxy industry. Additional applications include manufacturing photography development chemicals and cutting oils, bleaching activators for washing powders especially at low temperature, lubricant for plastics and polyamide process, and fuel additives. Diethylenetriamine is a yellow, hygroscopic liquid; boiling point of 206 C; soluble in water and hydrocarbons. It is used as a solvent for sulfur, acidic gas, resin and as a fuel and oil field component. It is used as an Intermediate for organic synthesis (modified polyamides, corrosion inhibitors, fuel additives, epoxy curing agents, fabric softeners and adhesion promoters) and saponification agent for acidic materials. Triethylenetetramine is a clear to yellowish oily liquid; melting point 12 C, boiling point 280 C. It is miscible with water and the solution is alkaline ( (pH 10 at 10% solution). It reacts with ketones, halogenated hydrocarbons, nitriles, epoxides, and with strong oxidants. Commercial triethylenetetramine is a mixture of linear TETA (typically 60%) and branched or cyclic TETA such as N,N'-Bis(2-aminoethyl)piperazine, N-[1-(2-piperazin-1-yl-ethyl)]ethane -1,2-diamine, tris-(2-aminoethyl)-amine. TETA and its derivatives are used as an epoxy curing agent. Their applications are similar to those of ethylenediamine and diethylenetriamine. End-uses of ethylenediamine family products include:
|
||