PRODUCT IDENTIFICATION
HS CODE
Oral ratLD50: 80 mg/kg
Chromic anhydride; Chromium anhydride;
Chromium VI oxide; Chromium trioxide anhydrous; Chromic trioxide; Chromerge; Chromic acid, solid; Chromium(VI) oxide (1:3); Anhydride chromique; Anidride cromica (Italian); Chrome (Trioxyde De) (French); Chrome Trioxydede; Chromia; Chromic (Vi) Acid; Chromsaeureanhydrid (German); Chromtrioxid (German); Chroomtrioxyde (Dutch); Chroomzuuranhydride (Dutch); Cromo(Triossido Di) (Italian); Chromium(VI) trioxide, Other RN: 12324-05-9, 12324-08-2
CLASSIFICATION
EXTRA NOTES
Poison/Deleterious Substance Code: 2-82
PHYSICAL AND CHEMICAL PROPERTIES
0.20-2.0 depending on concentration
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
EXTERNAL LINKS & GENERAL DESCRIPTION
Local: Chromium (symbol Cr and atomic number 24) occurs in the oxidation states 0, +2, +3, and +6 states. Element (0) and divalent chromium, however, are unstable. Chromium (0) immediately produce a thin oxide layer. Divalent chromium is easily oxidized to the trivalent form in air. The trivalent and hexavalent oxidation states are important in industry, which exit in their divalent anions called chromate and dichromate respectively and an chromic anhydride form called chromium trioxide (CrO3) and chromic oxide (Cr2O3). In industrial, chromium trioxide is called chromic acid. The principal uses of chromium are in the metallurgical processing of ferrochromium and other metallurgical products to impart corrosion resistance, chiefly stainless steel. There are applications in chrome plating, anodizing aluminium, and refractory processing of chrome brick. When combined with oxygen together other metallic elements such as lead and potassium, it forms various inorganic pigments. Chromium is used in chemical processing to produce chromic acid and chromates. Chromates are strong oxidants which will produce many organic and inorganic materials and used in the purification of chemicals. Chromates are used as rust and corrosion inhibitors in diesel engines. Dichromate is converted to chromic sulfate for tanning of leather. The reaction of chromium with collagen raises the hydrothermal stability of the leather and renders it resistant to bacterial attack. The reaction with collagen is useful reaction in screen printing application and in photography as a sensitizer for gelatin coatings. This Chromates and dichromates are used as pigments in paints and in dyeing. Chrome colors include black, red, orange, green, and yellow. Chromate salts contain the chromate ion, CrO4-2, and have an intense yellow color. Dichromate salts contain the dichromate ion, Cr2O7-2, and have an intense orange color. Chromates are used as mordant in dyeing cloth.
Chromic acid ( chromium trioxide, CrO3) is an odorless red deliquescent solid. Chromium trioxide is produced commercially by the reaction of sodium dichromate with concentrated sulfuric acid. It has been used mainly for chromium plating particularly in the production of automobiles and as a colorant in ceramics. Uses in other metal-finishing operations include aluminium anodizing, particularly on military aircraft; chemical conversion coatings, which provide both decoration and corrosion protection; and the production of phosphate films on galvanized iron or steel. It is a powerful oxidant and are utilized by controlled oxidations in organic synthesis. This compound is sensitive to moisture.
Another significant oxygen compound, chromic oxide is prepared by calcining sodium dichromate with boric acid or by reducing sodium dichromate with carbon. Anhydrous chromic oxide is produced commercially from chromic hydroxide, dry ammonium dichromate, or sodium dichromate by heating with sulfur. Chromic oxide is a dark green, amorphous powder, forming hexagonal crystals on heating that are insoluble in water or acids. Most chromic oxide is used as a pigment. Anhydrous chromic oxide is known as the most stable green pigment used when heat, light and chemical resistance is required for glass, ceramics, and polymers. Its hydrate form is called Guignet's green and used as a green pigment, particularly for automotive finishes.
Chromic compounds are also used in metallurgy in the manufacture of chromium metal and aluminium-chromium master alloys, in refractory brick, and as a chemical intermediate. They have good resistance to alkali and find application as colorant for latex paints. They are used in asphalt roofing and in camouflage paints. They are used as catalyst in the preparation of methanol, butadiene and high-density polyethylene. When used as a mild abrasive for polishing jewellery and fine metal parts, it is known as ��green rouge��
Chromic compounds are used extensively as pigments. Chromic acid finds applications in:
- wood Preservative
- Metal Plating
- Magnetic Tape
- Catalyst
- Copper stripping
- Aluminum anodizing
- Corrosion inhibitor
- Photography
- Purifying oil and acetylene
- Oxidizer in organic synthesis.
GENERAL DESCRIPTION OF CHROMATE OXIDIZER: Chromium does not occur by itself in nature but always in compounds. The stable oxidation states of chromium are +6, +3 (most stable), and +2. In industry, chromic acid refers to chromium(��) oxide or chromium trioxide (CrO3). This substance decomposes above 250 C to chromic oxide and oxygen. Chromium in the +6 (or ��) oxidation state (hexavalent chromium) is a strong oxidant and reacts violently with combustible and reducing materials. The solution in water is a strong acid, it reacts violently with bases and is corrosive. In organic synthesis, chromic acid is used to oxidize primary or secondary alcohols to aldehydes (or ketones) by oxidation state change from +6 to +3. It is difficult to stop at the aldehyde stage during the oxidation reaction, which usually proceeds to the carboxylic acid. Chromate oxidizers such as pyridinium chlorochromate are used to stop at the aldehyde group. The stable oxidation states +3 is the most stable. Saturated primary alcohols are oxidized to carboxylic acids in dimethylformamide solution. Jones reagent (sodium dichromate in aqueous sulfuric acid) continues the oxidation to the carboxylic acid product. Chlorochromates are efficient oxidizing agent for alcohols, allylic and benzylic methylene groups, and for oxidative cleavage of aryl substituted olefins. Hexavalent chromium solutions will not oxidize a tertiary alcohol. Chromium �� compounds have a dark orange to red color while chromium ��compounds are normally green. In medicinal field, they are used as anti-infective agents
APPEARANCE
Dark Red Flake
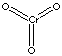
CrO3
99.5% min
SO4
0.15% max
WATER INSOLUBLES
0.01% max
Cl
0.01% max